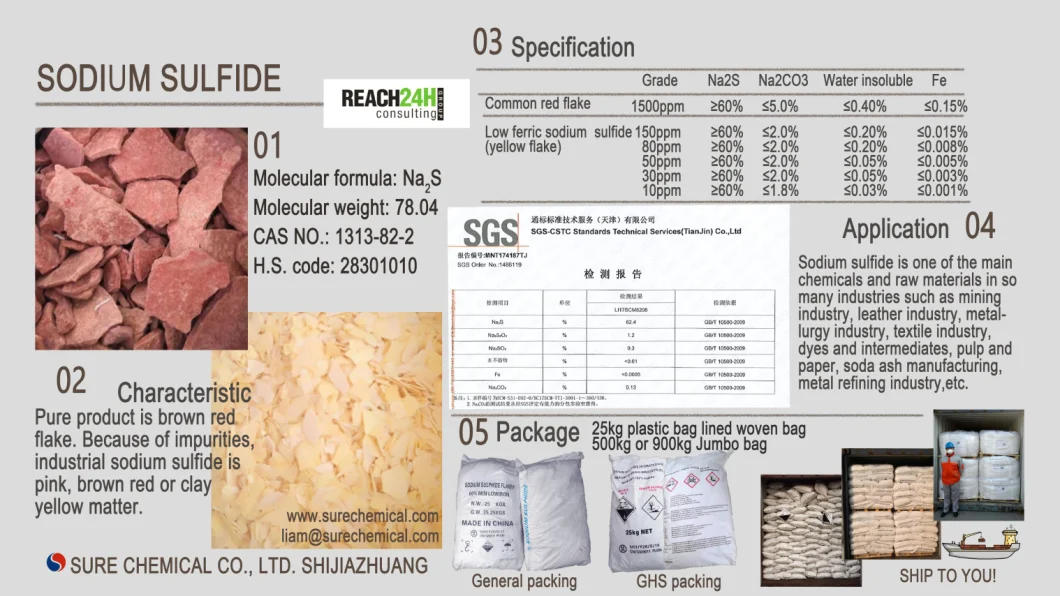
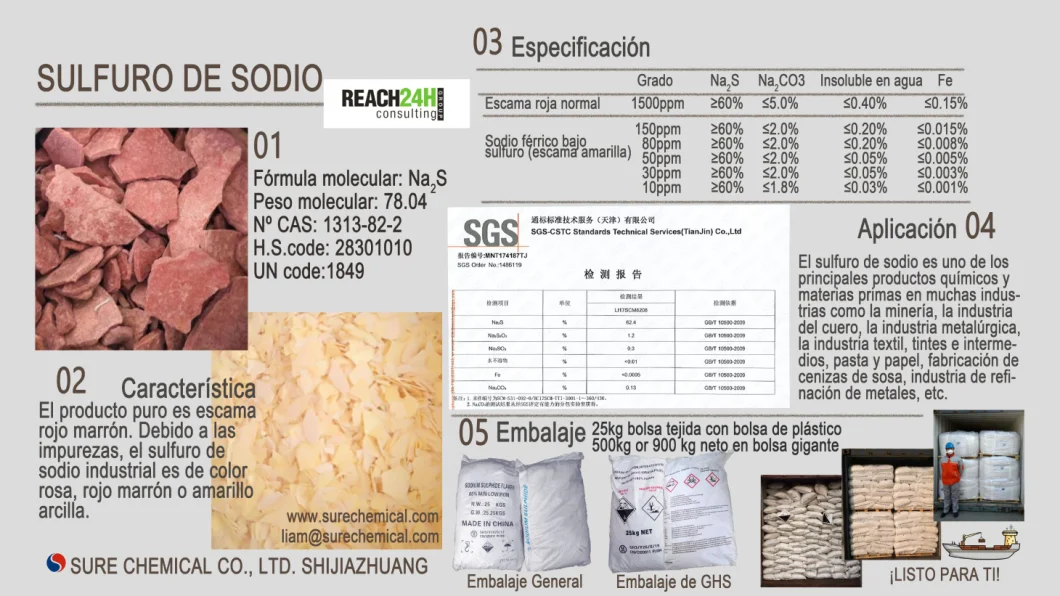
SODIUM SULFIDE
CAS NO. : 1313-82-2
Molecular formula: Na2S
Molecular weight: 78.04
UN code: 1849
How to buy Sodium Sulfide?
Characteristic: Pure product is brown red flake. Industrial sodium sulfide is pink, brown red or clay yellow matter due to impurities.
Specifications:
Application:
It can be used in sulfur dye, reductant of dye intermediate, mordant, flotation agent of ore, depilation agent of leather, paper cooking agent. Moreover, it is widely used in textile, pigment and rubber industry, or used in the production of sodium thiosulfate, sodium polysulfide and sodium hydrosulphide.
Package: 25kg plastic bag lined woven bag / 500kg or 900kg Jumbo bag
A good beginning is half the battle :First time Na2S from China export to EU market!
1fcl Sodium sulphide (low iron content grade) exported to Malaysia
The function of Sodium Sulfide in flotation
Mechanism:
1. Sulfide ore inhibitor
(1) When the HS-ion concentration reaches a certain value, competitive adsorption occurs on the mineral surface, and the HS-ion displaces the xanthate anions that have adsorbed on the mineral surface.
(2) The hydrophilic HS-ion itself is adsorbed on the surface of the sulfide ore, increasing the mineral's hydrophilicity, for the mineral is inhibited.
(3) The solubility product of the metal sulfide is smaller than the solubility product of the metal xanthate, and the S2-in the solution reacts with the xanthate anion, squeezing the xanthate down, so it is suppressed.
2. Activator of non-ferrous metal oxide ore
After the surface of the ore is sulfided with Na2S, a sulfided film is formed to activate the minerals, and flotation is carried out with xanthate collectors. Such as the sulfidation reaction of galena:
PbCO3] PbCO3+2Na2S=PbCO3] PbS+2Na2CO3
3. Desorb the collector on the surface of sulfide ore
As HS- and S2- ions have a strong affinity for sulfide minerals, they can have a strong adsorption on the mineral surface. As long as the concentration of these ions in the pulp is large enough, the adsorbed xanthate on the mineral surface can be desorbed.
The main functions:
1. Sodium sulfide is an inhibitor of most sulfide ores
When the amount is large, it can inhibit most of the sulfide ore. It inhibits the decreasing order of sulfide ore roughly: galena, sphalerite, chalcopyrite, porphyrite, copper blue, pyrite, chalcocite. As molybdenite is naturally floatable, sodium sulfide cannot suppress it. In that case, when floating molybdenite, Na2S can be used to suppress other sulfide ores.
2. Sodium sulfide is a vulcanizing agent for non-ferrous metal oxide ores
Nonferrous metal oxide ore cannot be directly captured by xanthate. However, if sodium sulfide is added before flotation with xanthate, a thin film of sulfide ore can be produced on the surface of the mineral, and xanthate can capture it. This effect of sodium sulfide is called sulfidation, so it can be used as a vulcanizing agent for non-ferrous metal oxide ores. After the white lead ore reacts with sodium sulfide, the surface color changes from white to dark. After the action of malachite and sodium sulfide, the surface color changes from green to dark, indicating that after vulcanization, sulfide films different from minerals are formed on the surfaces of these two minerals.
3. Sodium sulfide is a de-agent for mixed concentrate of sulfide ore
When the amount of sodium sulfide is large, it can resolve the xanthate collector that adsorbs on the surface of minerals. Therefore, sodium sulfide can be used as a de-agent before the separation of mixed concentrates. For example, before separation of lead-zinc mixed concentrate or copper-lead mixed concentrate, the slurry can be concentrated, a large amount of sodium sulfide is added to remove the drug, then washed, and fresh water is added to adjust the slurry for separation and flotation.
Sodium Sulfide (Na2S) is a commonly used agent in the flotation of sulfide ores. In addition to the above three main functions, sodium sulfide also has the effect of eliminating certain ions in the slurry that are harmful to flotation, because it can form insoluble sulfide precipitates with many metal ions.